Glycerol as building block for new synthesis
Summary of the work made
Two different original processes have been developed using glycerol as raw material for producing respectively di-chlorohydrins and the mixture of di- and tri-isobutyl ethers (GTBE). Di-chlorohydrins are important intermediates to produce epichlorohydrin used in the production of epoxy-resins. GTBE is used as fuels additive. The first process has been industrially realized with the partnership of CONSER SpA in China and proprietary patented catalysts are used . The second process gives as final product a mixture of biodiesel+ 7.5 % GTBE, because, biodiesel is used in the process as extracting agent of the reaction product GTBE. At this purpose, a detailed scheme of the process treating 500 Kg/h of glycerol has been published
Work
In the production of biodiesel, glycerol is obtained as by-product with a yield of 10% b.w. An excess of this substance is, therefore, available on the market and it becomes imperative to find new convenient uses for glycerol also in the perspective of reducing the cost of biodiesel production. A scheme of some possible reactions is reported below:
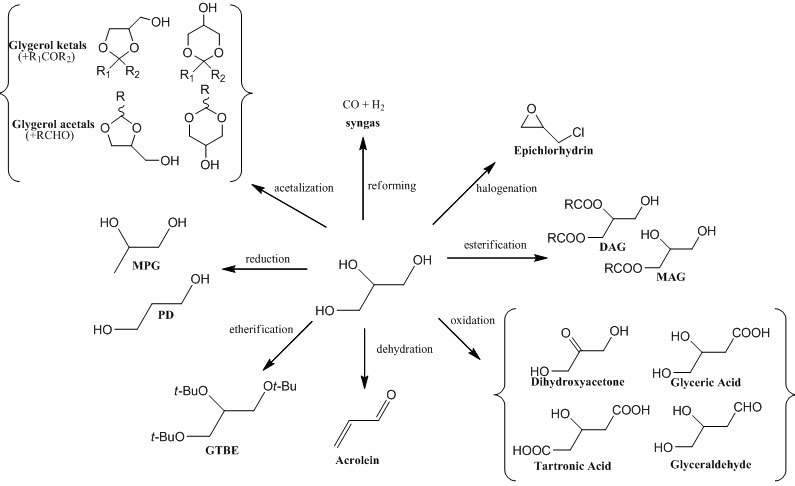
However, despite the large number of proposals, in practice, there are only two possible strategies to consume the large amount of glycerol deriving from biodiesel that are:
1) the use of glycerol as feedstock for obtaining commodities.
2) The use of glycerol for producing oxygenated additives for fuels.
Eurochem Engineering has developed two different processes in both the mentioned fields.
Glycerol hydrochlorination allows to obtain chlorohydrins, important intermediates for producing epichlorohydrin (a monomer of epoxide resins) through the following reaction steps:

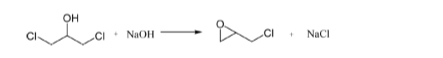
The hydrochlorination of glycerol is selective in giving 1,3 dichlorohydrin, while, starting from propene as in the traditional most practiced process a mixture of 1,2 and 1,3 dichlorohydrin (30:70%) is obtained. This is an advantage of the process via glycerol, because, 1,3 di-chlorohydrin is much more reactive than 1,2 di-chlorohydrin. Carboxylic acids are normally used as catalysts and proprietary catalysts have been developed by Eurochem Engineering srl. Many different works [1-5] and patents [6-7] have been published in collaboration with NICL and processes have been developed with the partnership of some other companies, in particular with CONSER SpA with which a collaboration agreement has been stipulated.
Different substances derived from glycerol can be used as blending components for diesel fuel such as: ethers (glycerol isobutylethers) , esters (triacetin), acetals and ketals (see previous scheme). However, the etherification to obtain di- and tri-isobutyl ethers (GTBE) is the most promising reaction. The target product is a mixture of the di- and tri- butyl-ethers of glycerin (h-GTBE), a good additive for diesel (both fossil and bio) and also for gasoline as octane-booster. In diesel and biodiesel (7.5% b.w.), it will lead to the reduction of the emissions of: particulates, NOx and unburned hydrocarbons. Moreover, blending diesel or biodiesel with h-GTBE reduces also: viscosity, cloud point and pour point. At this purpose, Eurochem Engineering developed a new process described in detail in [8].

-
Tesser, R.; Santacesaria, E.; Di Serio, M.; Di Nuzzi, G.; Fiandra, V. Kinetics of glycerol chlorination with hydrochloric acid: a new route to α,γ -dichlorohydrin. Industrial & Engineering Chemistry Research (2007), 46(20), 6456-6465.

-
Santacesaria, E.; Tesser, R.; Di Serio, M.; Casale, L.; Verde, D. New process for producing epichlorohydrin via glycerol chlorination; Industrial & Engineering Chemistry Research (2010), 49(3), 964-970.

-
R. Tesser, M. Di Serio, R. Vitiello, V. Russo, E. Ranieri, E. Speranza, E. Santacesaria; Glycerol Chlorination in Gas-Liquid Semibatch Reactor: An Alternative Route for Chlorohydrins Production; Industrial & Engineering Chemistry Research 51 (2012) 8768-8776.

-
R. Vitiello, V. Russo, R. Turco, R. Tesser, M. Di Serio, E. Santacesaria; Glycerol chlorination in a gas-liquid semibatch reactor: New catalysts for chlorohydrin production; Chinese Journal of Catalysis 35 (2014) 663–669

-
E. Santacesaria, R. Vitiello, R. Tesser, V.Russo, R.Turco, M. Di Serio; Chemical and Technical Aspects of the Synthesis of Chlorohydrins from Glycerol; Submitted for publication to Industrial & Engineering Chemistry Research (2014) 53, 8939-8962.

-
Santacesaria, Elio; Di Serio, Martino; Tesser, Riccardo. "Process for monochlorohydrins production from glycerol and hydrochloric acid” (EUROCHEM Engineering srl) PCT Int. Appl. (2008) WO 2008132770 A1 20081106
-
Siano, D.; Santacesaria, E.; Fiandra, V.; Tesser, R.; Di Nuzzi, G.;Di Serio, M.; Nastasi, M. (Eurochem Engineering srl). Process for the production of α,γ-dichlorohydrin from glycerin and hydrochloric acid. WO 111810 A2, 2006; U.S. Patent US 2009/0062574, 2009; Eur. Patent EP 1879842 B1, 2012; Chin. Patent ZL 2006, 80012864.9, 2006
-
Santacesaria Elio, Tesser Riccardo, Vitiello Rosa, Di Serio Martino; A new process for the production of di-chlorohydrins; It. Patent RM2013A000488; PCT/IT2014/000227
-
Di Serio M., Casale L., Tesser R., Santacesaria E. New Process for the Production of Glycerol tert-Butyl Ethers; Energy & Fuels (2010), 24(9), 4668-4672.
